With Cutting-edge Technologies

As participant in projects led by the Ministry of Science & Technology and China National Health Commission, we actively contribute to the prevention and control of infectious diseases.

We develop leading edge therapies for various cancers by combining next-generation In-silico Neoantigen Identification Methods with Built-in lentiviral vector-based platform.

As a CDMO, we offer effective solutions from clinical trials to commercialization, as well as delivery of safe and reliable viral vectors to accommodate our customers' needs.

Lentiviral vector technology stands as the culmination of over two decades of groundbreaking research at the renowned Pasteur Institute in France.

Jinwei's lentiviral vector products were utilized in a first-in human Phase I/II HIV clinical trial and have already been approved by FDA for upcoming HPV Phase I clinical trials.

Jinwei is dedicated to developing first-in-class innovative drugs using lentiviral vector technology platforms and cryptic peptide technology platforms, applicable to a wide range of infectious diseases and malignant tumors, to address the significant unmet clinical needs.

Partnership with the Huashan Hospital. Participant in projects led by the Ministry of Science & Technology and China National Health Commission.

State-of-the-art laboratory and GMP compliant production facility in accordance to main regulation authorities (FDA, EMA, CFDA and etc).
ABOUT US
Shanghai Jinwei Biotechnology Co., Ltd. (Jinwei) is a biotechnology company dedicated to pursue innovative technology to develop cures for people with serious and life-threatening diseases.
Lentiviral Vector
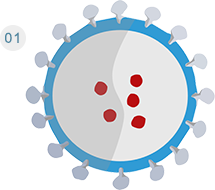
A lentiviral vector carries antigen genes from a pathogen or tumor cell.
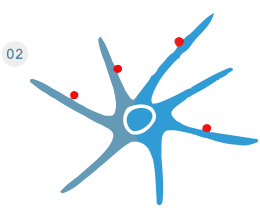
The lentiviral vector transcripts and releases the mRNA that encodes the antigen into the dendritic cell.
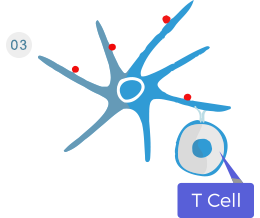
Dendritic cells presents the antigens to T cells.
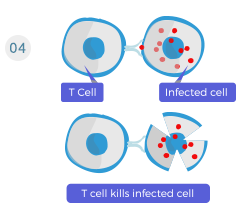
T cells recognize the antigens and kill the antigen-bearing cells (pathogens, infected or tumor cells).

Latest Update