

Authors: Calderón-Gómez, Lampropoulou V, Shen P, Neves P, Roch T, Stervbo U, Rutz S, Kühl AA, Heppner FL, Loddenkemper C, Anderton SM, Kanellopoulos JM, Charneau P, Fillatreau S
Activated B cells can regulate immunity and have been envisaged as a potential cell-based therapy for treating autoimmune diseases. However, activated human B cells can also propagate immune responses, and the effects resulting from their infusion into patients cannot be predicted. This led us to consider resting B cells, which in contrast are poorly immunogenic, as an alternative cellular platform for the suppression of unwanted immunity. Here, we report that resting B cells can be directly engineered with lentiviral vectors to express antigens in a remarkably simple, rapid, and effective way. Notably, this neither required nor induced activation of the B cells. With this approach we were able to produce reprogrammed resting B cells that inhibited antigen-specific CD4(+) T cells, CD8(+) T cells, and B cells upon adoptive transfer in mice. Furthermore, resting B cells engineered to ectopically express myelin oligodendrocyte glycoprotein antigen protected recipient mice from severe disability and demyelination in EAE, and even induced complete remission from disease in mice lacking functional natural Tregs, which otherwise developed chronic paralysis. In conclusion, our study introduces reprogrammed quiescent B cells as a novel tool for suppressing undesirable immunity.
Recent studies have identified novel roles of activated B cells in the inhibition of immunity [1-3]. These suppressive functions of B cells could be of interest to therapeutically block unwanted immune responses [1-3].
To suppress ongoing autoimmune diseases in mice B cells must be activated via Toll-like-receptors (TLR), B cell receptor for antigen (BCR), and CD40, which result in sustained production of the immunosuppressive cytokine interleukin(IL)-10 [2, 4]. These activation pathways may therefore be useful to produce therapeutic regulatory B cells. In keeping with this, B cells activated with selected TLR agonists in vitro effectively suppressed various autoimmune diseases upon adoptive transfer in recipient mice [5, 6]. The protective function of these activated B cells depended on presentation of disease-relevant autoantigens via major histocompatibility complex (MHC)-II molecules [5]. Importantly, activated B cells can also exert stimulatory activities, and the mechanisms distinguishing their pro- from anti-inflammatory functions have not been fully identified [7-10]. Thus, currently it is not possible to predict whether an ex vivo activated human B cell will mediate suppressive or stimulatory functions upon administration into patients. Consequently, the utilization of activated B cells may appear not safe enough for clinical application in humans. On the other hand, resting B cells could provide a safer platform for adoptive therapies: they express low levels of costimulatory molecules and they do not secrete antibodies [11]. Resting B cells can induce tolerance in naive T cells, but are less effective than some activated B cells in inhibiting ongoing immune reactions [11-13].
We therefore reasoned that the ideal therapeutic B cell should combine the weak immunogenicity of resting B cells with the effective suppressive functions of some activated B cells. In order to construct such “resting regulatory B cells”, we established a novel gene therapy protocol to genetically reprogram resting B cells while keeping them in a quiescent state. This enabled us to produce resting B cells presenting antigen to reactive T cells, and secreting the cytokine IL-10, which are two key features of suppressive activated B cells [1-3, 5]. We demonstrate that such reprogrammed resting B cells can suppress unwanted immune reactions mediated by CD4+ T cells, CD8+ T cells, and B cells upon adoptive transfer in recipient mice. These B cells protected recipient mice from chronic disability and demyelination in experimental autoimmune encephalomyelitis (EAE), a model of multiple sclerosis (MS). Altogether, our study introduces genetically reprogrammed resting B cells as a new vehicle for the inhibition of immunity.
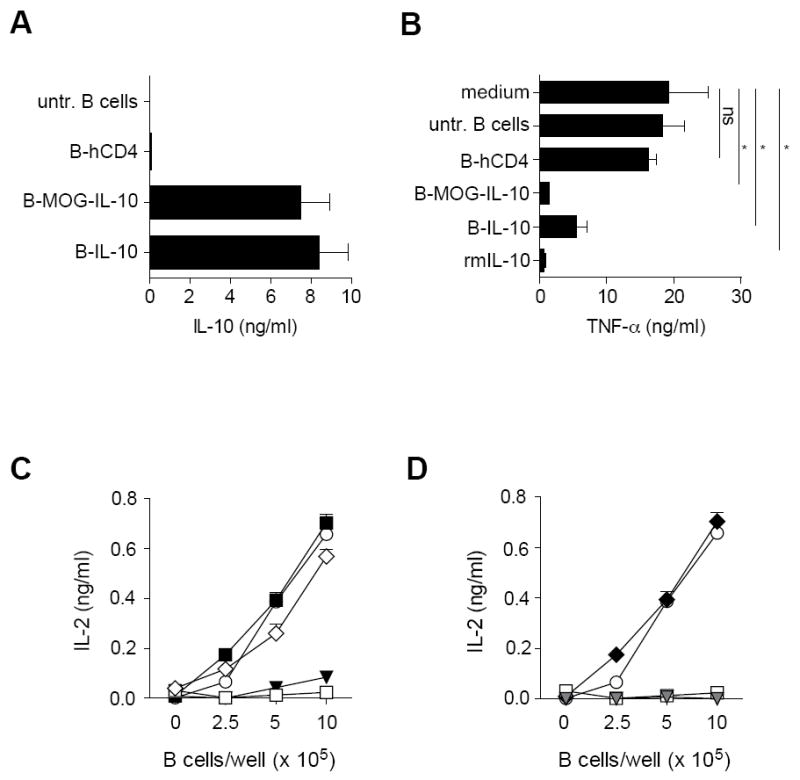
We established a lentiviral-based protocol to genetically reprogram quiescent B cells. Plain centrifugation of resting B cells with lentiviral particles for 75 minutes was sufficient to obtain high transduction efficiency (Fig. 1A).
Download from: http://europepmc.org/article/PMC/3431508
